From Contamination to Beautification: Ferns Remove Arsenic From Soil
For Students
Introduction
Arsenic is a chemical element that has the symbol As. Arsenic is widely distributed in nature and is associated with the ores of metals such as gold, lead, and copper. Arsenic also enters the environment through human activities, as arsenic is often used in pesticides, dyes, and chemical weapons. The arsenic compound chromated copper arsenate (CCA) has also been used as a wood preservative. At high doses, arsenic is a poison to humans and other animals which can cause death, and at lower doses over a longer time, it can also cause cancer. Arsenic contamination of soil and water is thus an environmental health issue.
In 2001, University of Florida researcher Lena Ma discovered that the Chinese brake fern (Pteris vittata) grows well in arsenic-contaminated soil. This plant accumulates large amounts of arsenic in its fronds, the portion of the fern that is above ground. This discovery led to the idea that brake fern could help clean up arsenic-contaminated soil. The use of plants to clean up contaminated soil and water is called phytoremediation. Phytoremediation offers an environmentally-friendly and cost-effective method to remove arsenic from contaminated soil. In addition to brake fern, other plants have been found to be useful in phytoremediation. Sunflowers were very effective at removing radioactive materials from the water near the site the 1986 nuclear-power-plant disaster at Chernobyl, Ukraine. Poplar trees are also useful in removing a wide range of pollutants from soil and have been used widely for this purpose.
The flowchart below illustrates how brake fern could be utilized in the remediation of arsenic-contaminated soil. It is important to note that the phytoremediation process does not result in the disappearance of the arsenic. During phytoremediation, the arsenic moves from the soil to the fern fronds. It is then easy to harvest the fern fronds and further concentrate the arsenic in a safer location. Sometimes remediation processes result in complete destruction of the contaminant, as when microorganisms degrade polyaromatic hydrocarbons completely to carbon dioxide and water.

Data Sets
These data sets are the results of experiments conducted by scientists who are investigating the Chinese brake fern and its application to phytoremediation of arsenic-contaminated soil.
A.
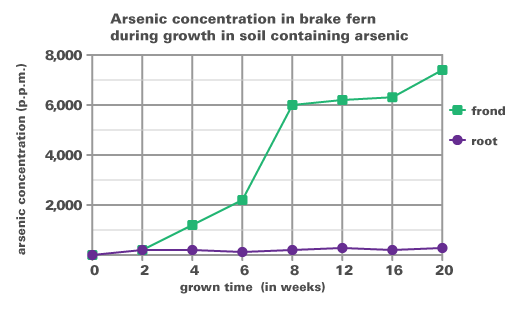
Method: Scientists collected brake fern plants from several uncontaminated sites and then planted them in pots containing soil. The soil contained 97 p.p.m. arsenic, and the brake fern plants were grown for 20 weeks. The concentration of arsenic was measured in the fern’s roots and fronds at various timepoints. The concentration of arsenic is expressed in p.p.m. The abbreviation p.p.m. stands for parts per million, or one milligram arsenic per kilogram of soil or plant.
Questions:
- Where does the fern concentrate the arsenic – in the frond or in the root?
- Why might the fern store the arsenic here?
- When did the fern show the greatest increase in frond arsenic concentration?
- How could you design an experiment to test this hypothesis?
- What is a hypothesis for this experiment?
B.
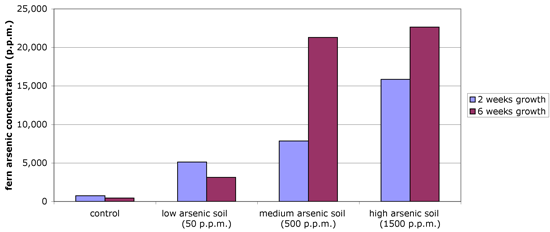
Method: In another experiment, brake ferns were planted in pots containing various amounts of arsenic (50 p.p.m., 500 p.p.m., and 1500 p.p.m.). The control soil contained no added arsenic, though very low levels of arsenic are naturally present in soil. After 6 weeks of growth, the scientists measured the amount of arsenic present in the fern frond.
Questions:
- Was the fern effective at removing the arsenic from the soil?
- Compare the 2 week and 6 week values for arsenic concentrations in the ferns. How does the arsenic-removing ability of the fern compare with low arsenic (control, low-As) versus higher arsenic (As-contaminated soil, medium As, high As) concentrations?
- Why was the control soil included?
- These data show the average arsenic content of four replicate ferns grown during the experiment. Why did the scientist use replicates and average the measurements from each replicate?
C.
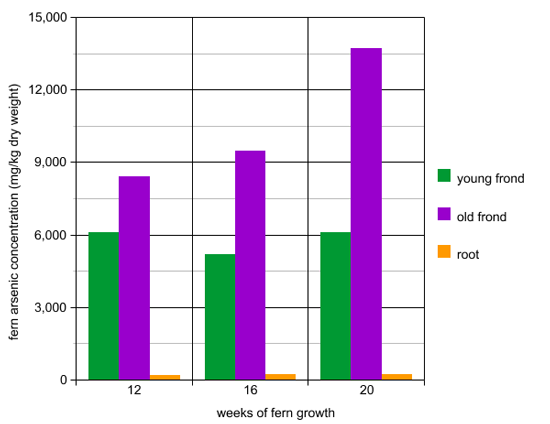
Method: In another experiment, ferns were planted in pots with arsenic-contaminated soil. After 12, 16, and 20 weeks of growth, the scientists measured the amount of arsenic present in the ferns. Specifically, they measured the arsenic concentrations in old fronds, young fronds, and the roots. Mg/kg dry weight is a concentration measurement that means mg arsenic per kg dry weight of fern.
Questions:
- Does the fern store arsenic in the root or in the frond?
- Does the concentration of arsenic in the young fronds change much between 12, 16, and 20 weeks of growth?
- Does the concentration of arsenic in the old fronds change much between 12, 16, and 20 weeks of growth?
D.
Grasshopper herbivory and arsenic-containing ferns.
Method: Grasshoppers are herbivourous predators, which means they graze on plants. Ferns are a food source for grasshoppers. Hungry grasshoppers were placed by themselves in small boxes. They were offered a small piece of fern frond as their only food source. The fronds were taken from ferns grown in arsenic-contaminated soil (experimental) or uncontaminated soil (control). The fern fronds were weighed daily for 5 days to determine how much fern the grasshopper ate. The data is presented below as a graph and in a table.
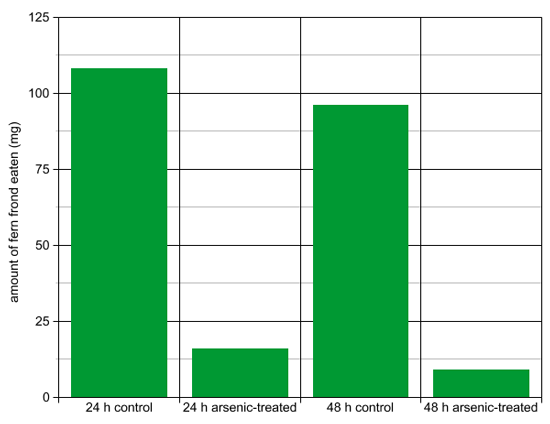
| Amount of fern frond eaten (mg) | |
|---|---|
| 24 h control | 108 |
| 24 h arsenic-treated | 16 |
| 48 h control | 96 |
| 48 h arsenic-treated | 9 |
Questions:
- What hypothesis might this experiment be designed to test?
- Does the amount of fern frond eaten change much between 24 and 48 h?
- Why might the scientist have chosen 24 and 48 hour timepoints to measure the amount of fern frond eaten?
- Did this experiment confirm or deny the hypothesis that “the fern’s accumulation of arsenic in its frond is a defense mechanism to avoid herbivorous predation”?
- Do the table and the graph present the same information?
- Which do you prefer for this data: the graph or the table?
- In general, when might you choose a graph instead of a table to present data? What factors influence the decision?
E.
Method: In a similar experiment, hungry grasshoppers were placed by themselves in small boxes. This time, they were offered a choice of three pieces of lettuce. The lettuce had been dipped in water for 10 seconds. One piece of lettuce was dipped in water containing a high concentration of arsenic (1.0 mM), one piece of lettuce was dipped in water containing a low concentration of arsenic (0.1 mM), and one piece of lettuce was dipped in water containing no arsenic (0 mM). After 24 h or 48 h, the amount of each piece of lettuce that had been eaten was scored on a scale of 1-10, with 10 representing a completely eaten lettuce leaf. The abbreviation mM stands for millimolar. One molar means one mole of a substance per liter of water.
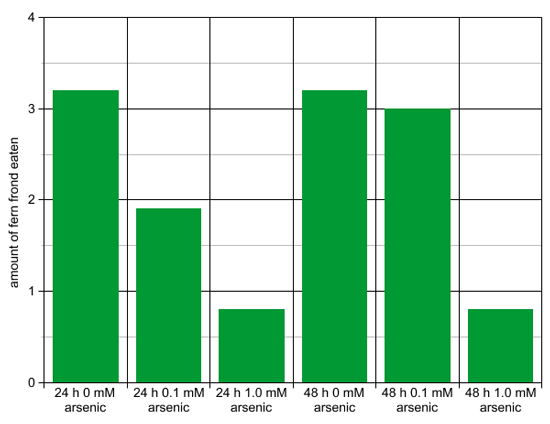
Questions:
- Was there any difference in the amount of control lettuce eaten between 24 and 48 h?
- Is this what you expect in a control?
- Did the grasshoppers eat any more of the 1.0 mM arsenic lettuce at 48 h than at 24 h?
- What does this tell you about the willingness of grasshoppers to eat lettuce dipped in 1.0 mM arsenic?
- Did the grasshoppers eat any more of the 0.1 mM arsenic lettuce at 48 h than at 24 h?
- What does this tell you about the willingness of grasshoppers to eat lettuce dipped in 0.1 mM arsenic?
- Why do you think the scientists used the 1-10 scale to score the amount of lettuce eaten? Why didn’t they just weigh the lettuce as they weighed the fern fronds in the previous experiment?
F.
Method: This experiment tested the effect of fertilizers on brake fern’s accumulation of arsenic. The three types of fertilizers tested were Compost 1, Compost 2, and phosphate rock. Brake fern plants were grown in pots in soil with 125 mg/kg arsenic. Equal amounts of fertilizer were added to experimental pots, and no fertilizer was added to the control pots. The scientists measured the arsenic concentration in the roots and fronds after 12 weeks of growth.
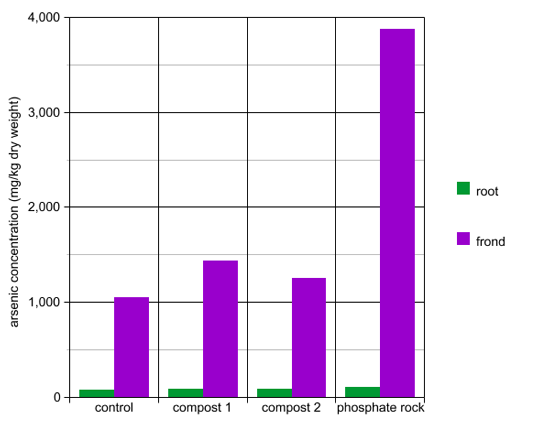
Questions:
- Did any fertilizer cause the fern to accumulate large amounts of arsenic in the root instead of the frond?
- Did application of the fertilizers increase arsenic uptake in the ferns?
- Which fertilizer was the most effective at increasing fern arsenic uptake?
- Which fertilizer would be the best choice to add to ferns when planning a phytoremediation project?
- What other factors might influence the decision of which fertilizer to choose?
G:
Brake fern distribution
View a map of brake fern distribution in Florida.
Why might the fern be present in some counties but not in others?
Extension activity:
Fern phytoremediation of arsenic-contaminated soil
Here are some values you can use to predict how ferns might be used to phytoremediate an arsenic-contaminated soil.
- Typical arsenic concentration in contaminated soil = 100 p.p.m. = 100 mg arsenic per kg soil
- One brake fern plant can extract 38 mg arsenic from soil in 20 weeks
- 90% of the extracted arsenic is stored in the frond (aboveground biomass)
Assume that ferns can remove 20 mg arsenic per kg soil in 20 weeks. At the end of 20 weeks, ferns are harvested and replanted until the desired soil arsenic concentration is reached. How many harvests would be required to phytoremediate a soil contaminated with 100 mg/kg arsenic?
How long would this take?
How many harvests would be required to phytoremediate a soil contaminated with 400 mg/kg arsenic?
How long would this take?
Is it realistic to assume that the ferns will always be able to remove 20 mg arsenic per kg soil in 20 weeks?
Why or why not?
What might be some limitations of using brake ferns to phytoremediate arsenic-contaminated soil?
Do research to find an arsenic-contaminated site that interests you. Using the information here and other data you find in your research, make a plan to use brake ferns to phytoremediate the site. What is the history of the site? How big is it? Where is it located? Is it contaminated with compounds other than arsenic? How many fern plants will be needed? How long will the phytoremediation take? What will be done with the land after the cleanup is complete?
Other phytoremediation applications:
- Salt-tolerant barley and/or sugar beets are commonly used for the extraction of sodium chloride (table salt) to reclaim fields that were previously flooded by sea water.
- Sunflowers were very effective at removing radioactive materials from the water near the site the 1986 nuclear-power-plant disaster at Chernobyl, Ukraine.
- Poplar trees are also useful in removing a wide range of pollutants from soil, from pesticides to heavy metals. Poplar trees are good candidates for phytoremediation.
Research one of these topics (or another topic of your choice) further online.
Web site resources
- Create A Graph, from the US Department of Education's National Center for Educational Statistics. This outstanding web interface allows students to create several different types of graphs. Highly recommended. Free!
- InspireData, commercially available graphing software
- web site for Dr. Lena Ma, the scientist who discovered the arsenic-accumulating properties of brake fern
- Soil contamination and bioremediation information
- Data used in this activity was obtained from the following publications. The articles can be downloaded from the following site: http://lqma.ifas.ufl.edu/PUBLICATION.html
- Cao, X., L.Q. Ma, and A. Shiralipour. 2003. Effects of compost and phosphate amendments on arsenic leachability in soils and arsenic uptake by Chinese Brake (Pteris Vittata L.) Environ. Pollution. 126:157-167.
- Ma, L.Q., K.M. Komar, C. Tu, W. Zhang,and Y Cai. 2001. A fern that hyperaccumulates arsenic. Nature. 409:579.
- Rathinasabapathi B, L.Q. Ma, and M. Srivastava. 2006. Arsenic hyperaccumulating ferns and their application to phytoremediation of arsenic contaminated sites. In: Floriculture, Ornamental and Plant Biotechnology: Advances and Topical Issues (1st Edition), Teixeira da Silva JA (ed), Global Science Books, London, UK, pp 305-311.
- Rathinasabapathi, B., M. Rangasamy, J. Froeba, R. H. Cherry, H. J. McAuslane, J.L. Capinera, M. Srivastava, & L.Q. Ma. 2007. Arsenic hyperaccumulation in the Chinese brake fern (Pteris vittata) deters grasshopper (Schistocerca americana) herbivory. New Phytologist. 175: 363–369.
- Tu, C., L.Q. Ma, and B. Bondada. 2002. Arsenic Accumulation in the Hyperaccumulator Chinese Brake Fern (Pteris vittata L.) and Its Utilization Potential for Phytoremediation. J. Environ. Qual. 31:1671-1675.